Abstract
Background: Autologous stem cell transplantation (ASCT) in multiple myeloma (MM) has been shown to improve survival compared to conventional chemotherapy alone. However, the ability to perform ASCT relies, in part, on collecting a sufficient number (#) of CD34+ hematopoietic stem cells (HSCs), typically from peripheral blood. The ideal HSC mobilization regimen would enable collection of optimal #s of HSCs (5-6x10 6 CD34+ cells/kg) within the minimum # of apheresis sessions possible. Yet, despite currently available G-CSF (G) based mobilization regimens and multiple apheresis days, many remain unable to collect optimal #s of HSCs. Motixafortide (M) is a novel CXCR4 inhibitor, with high affinity (IC 50 0.54-4.5 nM) and long receptor occupancy (>48 hours).
Methods: In this prospective, phase 3, double blind, placebo controlled, multicenter trial, 122 patients were randomized (2:1) to receive either M+G or placebo (P)+G for HSC mobilization prior to ASCT for MM. All patients received G (10 mcg/kg) on days 1-5 (and 6-8, if needed). Patients received either M (1.25 mg/kg, subcutaneous injection) or P on day 4 (and 6, if needed). Apheresis began day 5, with the primary (PEP) and secondary (SEP) endpoints of collecting ≥6x10 6 CD34+ cells/kg in up to 2 apheresis days or 1 day, respectively. Apheresis continued on days 6-8 if needed. Total CD34+ cells/kg were analyzed on site to determine if patients mobilized to the goal and all samples were subsequently sent for assessment by central laboratory. Patients that did not collect ≥2x10 6 CD34+ cells/kg by day 8 proceeded to rescue mobilization. The # of CD34+ cells infused was determined independently by each investigator according to local practice (minimum ≥2x10 6 CD34+ cells/kg). Analyses of the PEP/SEPs were performed on an intent-to-treat basis.
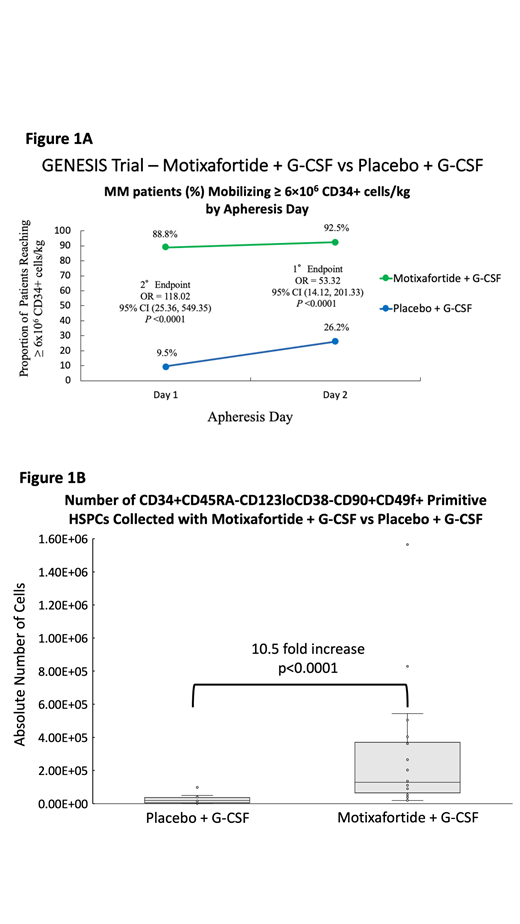
Results: Demographics between the 2 treatment arms were similar. Mobilization with M+G resulted in 92.5% of patients collecting ≥6x10 6 CD34+ cells/kg within 2 apheresis days vs 26.2% with P+G (Odds Ratio (OR) 53.3, 95% CI 14.12-201.33, p<0.0001). Furthermore, 88.8% of patients with M+G collected ≥6x10 6 CD34+ cells/kg in 1 apheresis day vs 9.5% with P+G (OR 118.0, 95% CI 25.36-549.35, p<0.0001); and 96.3% with M+G collected ≥2x10 6 CD34+ cells/kg within 1 apheresis day vs 64.3% with P+G (OR 18.9, 95% CI 4.47-80.04, p<0.0001). The PEP and SEPs were confirmed as statistically significant by central laboratory (all respective p-values <0.0001). The median # of HSCs mobilized in 1 apheresis day with M+G was 10.80x10 6 CD34+ cells/kg vs 2.14x10 6 CD34+ cells/kg with P+G. The # of cells infused was determined independently by each investigator according to local practice. Median time to neutrophil engraftment was 12 days in both arms (HR 0.94, 95% CI 0.62-1.41, p=0.75). Median time to platelet engraftment was 18 days (range: 17-19) with M+G and 17 days (range: 17-18) with P+G (HR 0.89, 95% CI 0.59-1.34, p=0.57). Graft durability at day 100 post-ASCT was 92.2% in the M+G arm and 91.9% in the P+G arm (OR 1.04, 95% CI 0.2-4.5, p=0.96). Overall, adverse events were reported in 98.8% (Grade 3/4: 68.8%) of patients with M+G vs 95.2% (Grade 3/4: 42.9%) with P+G, with cytopenias in the post-ASCT period prior to engraftment accounting for the majority of Grade 3/4 AEs in both arms, as expected. The most common AEs related to M included: local injection site reactions (any grade: 70.0%, Grade 3/4: 11.3%); and systemic reactions such as pruritis (33.8%), flushing (32.5%) and urticaria (12.5%). Additionally, mobilization with M+G resulted in a 10.5x increase in mean absolute # of CD34+CD45RA-CD123loCD38-CD90+CD49f+ primitive HSCs collected vs P+G (p<0.0001). Multicolor FACS and scRNA sequencing of CD34+ HSCs from both arms will be reported in a separate ASH abstract.
Conclusions: A single injection of M on top of G significantly increased the proportion of patients mobilizing ≥6x10 6 CD34+ cells/kg for ASCT (92.5%) vs G (26.2%) in up to 2 apheresis days (p<0.0001), while enabling 88.8% to collect ≥6x10 6 CD34+ cells/kg in just 1 apheresis (p<0.0001, Figure 1A). Despite the higher # of cells mobilized by M+G, the # of CD34+ cells/kg infused was determined independently by each investigator according to local practice with comparable engraftment kinetics and graft durability between the 2 arms. Finally, M+G mobilized 10.5x more immunophenotypically primitive CD34+ HSCs capable of durable multilineage hematopoietic engraftment vs P+G (p<0.0001, Figure 1B).
Crees: BioLineRx Ltd.: Research Funding. Larson: TORL biotherapeutics: Current holder of individual stocks in a privately-held company; Bioline: Research Funding; Abbvie: Research Funding; BMS: Research Funding; Celgene: Research Funding; GSK: Research Funding; Janssen: Research Funding; Juno: Research Funding; Novartis: Research Funding; Pfizer: Research Funding; Takeda: Research Funding. Illés: Pfizer: Consultancy, Membership on an entity's Board of Directors or advisory committees; Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees; Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees; Roche: Consultancy, Membership on an entity's Board of Directors or advisory committees; Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees. Stiff: Incyte: Research Funding; Cellectar: Research Funding; Seagen: Research Funding; Gamida Cell: Research Funding; Cellectar: Research Funding; Actinium: Research Funding; Bristol Myers Squibb: Research Funding; BioLineRX: Research Funding; Macrogenics: Research Funding; CRISPR Therapeutics: Consultancy, Honoraria; Amgen: Research Funding; Janssen: Research Funding; Kite, a Gilead Company: Research Funding; Karyopharm: Consultancy, Honoraria; MorphoSys: Consultancy, Honoraria. Sborov: Janssen: Consultancy, Membership on an entity's Board of Directors or advisory committees; GlaxoSmithKline: Consultancy; Sanofi: Consultancy; SkylineDx: Consultancy. Pereira: Jazz Pharmaceutical: Membership on an entity's Board of Directors or advisory committees. Mikala: Novartis: Consultancy; Takeda: Consultancy; Abbvie: Consultancy; Krka: Consultancy; Janssen: Consultancy; Amgen: Consultancy; Celgene: Consultancy. Holtick: Celgene: Honoraria; Sanofi: Honoraria. Qazilbash: Amgen: Research Funding; Oncopeptides: Other: Advisory Board; Bristol-Myers Squibb: Other: Advisory Board; Biolline: Research Funding; Angiocrine: Research Funding; NexImmune: Research Funding; Janssen: Research Funding. Hardy: American Gene Technologies, International: Membership on an entity's Board of Directors or advisory committees; Kite/Gilead: Membership on an entity's Board of Directors or advisory committees; InCyte: Membership on an entity's Board of Directors or advisory committees. Vainstein: BioLineRx LTD: Current Employment. Sorani: BioLineRx LTD: Current Employment. Gliko-Kabir: BioLineRx Ltd.: Current Employment. Goldstein: BioLineRx Ltd.: Current Employment. Kadosh: BioLineRx Ltd.: Current Employment.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal